EFSA to assess BSE risk posed by the use of ruminant collagen and gelatine in feed for pigs and poultry

EFSA’s Scientific Panel on Biological Hazards (BIOHAZ panel) has agreed to carry out the risk assessment. The EU risk assessor’s scientific opinion on the issue is due at the end of September 2020.
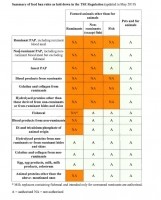
Currently, foodstuffs containing non-ruminant collagen and/or gelatine, which are no longer intended for human consumption but do not pose any public or animal health risk, are permitted for feeding to farmed animals in the EU. Foodstuffs containing ruminant collagen and/or gelatine - from both hides and skins and bones - are still banned, except in the case of food for pets and fur animals.
Only hydrolyzed proteins obtained from hides and skins of ruminant origin are authorized to be used in feed in the EU.
An estimated 100,000 tons of foodstuffs containing ruminant collagen and/or gelatine currently go for disposal and are thus underused within the EU, said the Commission in its letter to EFSA seeking the risk evaluation.
Industry, it said, is asking for the lifting of the current feed ban on edible ruminant collagen/gelatine (including those obtained from bones other than specified risk materials as defined in Article 3(l) (g) of Regulation (EC) No 999/2001) for non-ruminant farmed animals.
Commenting on the development, Arnaud Bouxin, deputy secretary general, FEFAC, told us:
“A lifting of the ban on the use of collagen and gelatin from ruminants in feed for non-ruminant farmed animals would mean [industry] being able to use more former foodstuffs containing these ingredients, thus avoiding [such material being] discarded as waste. But, of course, guarantees of safety are essential. We welcome the decision of the EU Commission to ask EFSA to perform a risk assessment.”
Background
The risk assessment request was made in the context of the Commission’s Communication to the European Parliament and the Council known as the TSE Road Map 2, a strategy paper on Transmissible Spongiform Encephalopathies (TSEs) for 2010-2015, which was adopted in July 2010. It outlines areas where future possible changes to EU legislation on TSEs could be made, with plans to review the existing feed ban rules laid down in EU legislation.
The ultimate purpose of the feed ban was to prevent the recycling of the BSE agent in cattle in the feed chain, and the subsequent risk of transmission to humans causing Variant Creutzfeldt-Jacob Disease (vCJD).
Therefore, the control system laid down in Annex IV to Regulation (EC) No 999/2001 was created to minimize presence of prions in feedstuffs and, ultimately, food. In addition, Regulation (EC) No 1069/2009 and its Implementing Regulation (EU) No 142/2011 laid down the conditions for the production of gelatine and collagen for its use in feed.