FDA sees drop for in-feed antimicrobial sales

The US Food and Drug Administration (FDA) released details of total antimicrobial sales for use in feed or with food-producing animals for 2017 in a report released at the end of December. It showed a second year of declining sales.
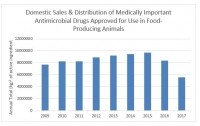
In total, the sales and distribution of medically important antimicrobials for use with food-producing animals fell by 33% from 2016 to 2017 and total sales fell 41% from 2015 to 2017, the FDA reported.
However, sales data does not necessarily indicate the actual use of antimicrobials.
The report is the first to include sales data following the implementation of the FDA’s guidance for industry 213, which addressed production uses of antimicrobials considered medical important for humans and the Veterinary Feed Directive (VFD) regulations requiring veterinary approval for medically important antimicrobial products.
The FDA’s goal with actions including the guidance provided and the Center for Veterinary Medicine’s action plan is to “support” the use of “good antimicrobial stewardship practices” and hinder the expansion of bacteria resistant to antimicrobials, not to lower sales, said an FDA spokesperson.
“The FDA’s actions over the last several years, including Guidance for Industry (GFI) #213 and #209 (judicious use) have fundamentally changed the way animal producers obtain and use medically important antimicrobials in food animals,” she told FeedNavigator. “The substantial reduction in the quantity of such drugs sold or distributed in 2017 demonstrates that ongoing stewardship efforts, including those initiated by FDA and other key stakeholders, are having a measurable impact.”
“Optimizing how medically important antimicrobial drugs are used and limiting their use to only when necessary to treat, control, or prevent disease will help to preserve the effectiveness of these drugs for fighting disease in both humans and animals,” she added.
The report is part of an ongoing effort to address a reporting requirement put in place by Congress in 2008, the spokesperson said.
A goal of the project was to support the FDA’s continuing analysis of the “interactions (including antimicrobial resistance), efficacy, and safety of antimicrobials approved for use in both humans and food-producing animals."
Additionally, in 2019, the FDA plans to publish a report looking at sales and distribution data along with other data sets to “more fully assess the progress of antimicrobial stewardship efforts,” she said.
Antimicrobial sales highlights
The total amount of antimicrobials sold for use in feed or with food-producing animals in 2017 was about 10.93m kg, the FDA said.
Within that amount, about 51% or 5.56m kg were medically important and 49% or 5.37m kg were not medically important.
The types of medically important antimicrobials most often sold for use with food-producing animals in 2017 were tetracyclines and aminoglycosides, the agency said.
“Tetracyclines accounted for 64%, penicillins for 12%, macrolides for 8%, sulfas for 5%, aminoglycosides for 5%, lincosamides for 3%, and cephalosporins and fluoroquinolones each for less than 1%,” it added.
The only drug types to see increased sales from 2016 to 2017 were fluoroquinolones and lincosamides, the agency said.
The amount of sales of tetracyclines and aminoglycosides dropped 40% and 19% during that time period, respectively, while the total sales of not medically important ionophores declined 6%.
The majority, estimated at about 42%, of the medically important antimicrobials sold were intended for use in cattle, however, the total amount sold dropped 35% from the previous year, the FDA said.
In 2017, an estimated 36% of sales were focused on swine and about 5% for chicken production.
None of the major production species saw an increase in medically important antimicrobial sales, the agency reported.
Sales of products for chickens saw the largest percentage drop of about 47% from 508,800kg in 2016 to 268,047kg in 2017.
“An estimated 80% of cephalosporins, 72% of sulfas, 48% of aminoglycocides, and 44% of tetracyclines were intended for use in cattle,” the FDA said. “An estimated 84% of lincosamides and 40% of macrolides were intended for use in swine. An estimated 61% of penicillins were intended for use in turkeys.”
The majority of non-medically important antimicrobial products sold in 2017 were for use in cattle, about 3.14m kg, or chickens, 1.48m kg, the agency said.
Of the medically important antimicrobials sold in 2017, about 62% or 3.43m kg were intended for use in feed and about 30% of the amount sold could be added to water, the agency said.
Sales of in-feed antimicrobials dropped 40% from 2009 to 2017 and 43% from 2016 to 2017.