US sees uptick in sales of medically important antibiotics used in feed
The US Food and Drug Administration (FDA) released details about the amounts of antimicrobials sold or distributed in 2018 for use with livestock or food-producing animals on Tuesday [December 10].
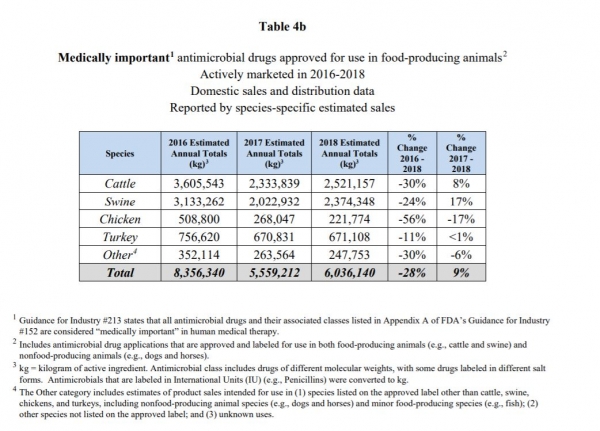
A key finding was that domestic sales and distribution of medically important antimicrobials approved for use in farmed animals increased by 9% from 2017 through 2018. Tetracyclines, which represent the largest volume of these domestic sales (3,974,179 kg in 2018), increased by 12% from 2017 through 2018.
The FDA has taken several steps in recent years including publishing new guidance for industry regarding the judicious use of medically important antimicrobials as part of its ongoing five-year plan to alter how producers buy and use medically important antimicrobials, a spokesperson with the FDA told FeedNavigator.
Although there was an increase in the amount of medically important antimicrobials sold, the indication is that efforts to support antimicrobial stewardship are producing a positive effect, the FDA said.
The primary goal of work to improve stewardship is not focused on reducing the sales volume of antimicrobials, a FDA spokesperson said. It is “instead to support the implementation of good antimicrobial stewardship practices in order to slow the development of antimicrobial resistance.”
“Despite the slight increase in sales in 2018, the substantial reduction in the quantity of antimicrobial drugs sold or distributed in 2017 demonstrates that ongoing stewardship efforts… are having an impact,” she said. “Precisely measuring that impact is a challenging work in progress because sales volume alone is not a yardstick by which we can measure the impact of stewardship efforts. Additional sources of information including actual use data, animal demographic information, animal health data and data on resistance, should be considered when assessing the progress of efforts to combat antimicrobial resistance.”
Currently, the agency is working with partners in industry and academia to gather more information about “how, when, and why animal producers and veterinarians use medically important antimicrobial drugs in food-producing animals,” she said. There are plans to release a report integrating and analyzing these data in 2020.
Antibiotic sales data - a deeper dive
From 2017 to 2018, the total sale of medically important antimicrobials intended for use with livestock jumped, with products for cattle seeing an 8% increase in sales, while those for swine saw a 17% increase. Sales of antimicrobials intended to be used with turkey saw an increase of less than 1% and chicken a drop of 17%, the agency said.
About 3,862,586kg of antimicrobials were sold for use in feed – accounting for 64% of the medically important antimicrobials sold, the FDA said.
The sale of tetracyclines – which make up the largest volume of domestic sales – grew by 12% year-on-year, the agency said. Sales of tetracyclines accounted for about 66% of total sales, followed by penicillins at 12%, macrolides at 8%, sufas and aminoglycosides at 5%, lincosamides at 2%, cephalosporins at 1% and fluoroquinolones at less than 1%.
Across all the antimicrobials, about 42% of those sold were intended for use in cattle, about 39% in swine, 11% in turkeys and 4% for use with chickens, the agency said. “An estimated 81% of cephalosporins, 67% of sulfas, 47% of aminoglycocides, and 44% of tetracyclines were intended for use in cattle [and] an estimated 83% of lincosamides and 41% of macrolides were intended for use in swine,” it added.
The only types of antimicrobials that saw a decline in sales from 2017 to 2018 were lincosamides and the category not individually reported, the agency said. With a 13% increase, aminoglycosides saw the largest growth in sales from year to year.
Data needs, remaining questions
In addition to the sales data, the FDA is in the process of collecting some on-farm use data, the spokesperson said. However, the project is “resource-intensive” and does raise some questions regarding privacy.
The FDA’s Center for Veterinary Medicine, working with university researchers, has collected data on the use of antimicrobials with cattle, swine and poultry – the initial report from that project was released earlier this year and reports on swine and cattle are expected in 2020.
“FDA has been working closely with the US Department of Agriculture, providing input on recent surveys administered under the National Animal Health Monitoring System to collect information on antimicrobial use on farms,” she added.
However, the FDA is not the only organization interested in having more information about when, how and for how long medically important antimicrobials are being used with livestock.
The Pew Charitable Trusts have been seeking additional information regarding the use of medically important antibiotics with livestock as part of the organization’s focus on antibiotic resistance, said Karin Hoelzer, senior officer in health programs with Pew.
“The biggest concern is that we are facing the post-antibiotic era where antibiotics will have lost their efficacy,” she told us. “Every time that an antibiotic is used there’s a risk that resistance emerges - what that means is there’s a need to be good stewards of antibiotics.”
The released sales data was “not good news,” Hoelzer said. “We’d hoped to see further progress in reducing the need for antibiotics and the sales of antibiotics.”
One challenge is that although there is information on antibiotic sales, there is little data to help explain what could be driving that change, which makes it hard to “diagnose the problem,” she said.
“It certainly emphasizes that there’s an urgent need for the other stewardship efforts that FDA is planning to do in the next five years,” she said. “But it would be really helpful to better understand what really drove that decrease in sales in 2016 and 2017 and what drove the recent uptick.”
There would be several areas where additional data or information could be gathered including the implementation of a method to assess sales based on the biomass of production animals, she said. “FDA has suggested a method, had a draft method published two years ago but still hasn’t been finalized, which means that the methodology isn’t available to be applied to the sales data – it wouldn’t answer all the questions we have, but at least it would make the data more useful,” she added.
“One of the concern trends that we’d love to have better data to better understand is there seems to be an uptick in the use of certain antibiotics that are more important to human health like aminoglycosides, like penicillins,” Hoelzer said. “Understanding that some antibiotics are even more important to human health than others and seeing that uptick and not understanding what is driving that is concerning.”
Similarly, the increase in the in-feed use of antibiotics, without information to understand what could be prompting that practice, is a worry, she added.
Having more granular information regarding the challenges prompting antibiotic use, especially in cattle and swine, also could support research into alternative methods, like vaccines, probiotic use or new management practices, to manage those diseases, she said.
“It’s very hard to do that for all diseases at the same time given research limitations,” she said. “Having an idea of what is driving this antibiotic use helps [us] prioritize.”