Promotional Features
7 common questions about antibiotic use and poultry production
By Dr. Ellen Hambrecht and Barbara Brutsaert, DVM
Programme Management Poultry Gut Health, Trouw Nutrition
Antibiotic resistance is one of today’s most pressing global public health threats. Unchecked, antibiotic resistance could become the primary cause of human deaths by 2050. The severity of this problem has drawn global attention to the use and overuse of antibiotics in livestock production and the potential implications for antibiotic resistance in humans and animals. As the poultry industry responds with actions to reduce antibiotic use, now is a good time to address seven often-asked questions.
1. What is antibiotic resistance?
In this article’s context, antibiotic resistance refers to adaptations that may occur in bacteria exposed to antibiotics. As these microorganisms evolve, more of them may become resistant to traditional interventions such as antibiotics used to treat illnesses in humans and/or animals. Bacteria can become resistant to one or several different antibiotic classes. In the latter case, limited treatment options mean a patient could die from a simple infection such as a urinary tract infection. While antibiotic-resistance may occur naturally, it is encouraged in the presence of antibiotics. Antibiotic-resistant bacteria may spread between people and animals directly or through the environment, and from person to person. Worryingly, some resistance genes are located on mobile genetic elements (plasmids) and may be transferred from one bacterial species to another (e.g. from E. coli to Salmonella bacteria).
2. Is there a link between antibiotic use in livestock production and antibiotic resistance in human health care?
Evidence shows an association between reduced antibiotic use in food-producing animals and a reduction of antibiotic-resistant bacteria in these animals. This is also true for humans in direct contact with these animals, such as farmers. For the general human population, limited data prevents drawing firm conclusions, especially concerning the magnitude of the effect. However, extensive research into mechanisms of antibiotic resistance suggests antibiotic-resistant bacteria may spread from food-producing animals to their environment and to humans. Consequently, in November 2017, the WHO recommended farmers and the livestock industry stop using antibiotics for the purposes of growth promotion and prevention of diseases not clinically diagnosed.
3. What about ionophores used to control coccidiosis?
Though classified as antibiotics, ionophores are often addressed separately with regards to reducing antibiotic use in poultry production. This speaks to the economic importance and significant animal welfare implications of coccidiosis, both in itself and as a predisposing factor for other problems such as necrotic enteritis. Vaccination with virulent or attenuated live strains of coccidia is an alternative increasingly applied in some regions. However, vaccination is more complex and more prone to management mistakes, which may explain to some extent the varying degrees of effectiveness. Vaccination also is generally more expensive than coccidiosis control by anti-coccidials, depending on the type of vaccine registered (virulent or attenuated live strains) and the extent of necrotic enteritis that is otherwise, as a side effect, controlled by ionophores.
Ionophores are not important in human health care. As such, resistance developed against ionophores is not relevant for human health. This raises a question as to why the U.S. FDA, for example, includes ionophores in its definition of antibiotics used in livestock production. First, ionophores are by definition antibiotics. Beyond their effect on coccidia, ionophores exert an antibiotic effect on Clostridium bacteria. A second consideration is consumer trust. Not labeling ionophores as antibiotics could confuse consumers and undermine trust in production schemes such as “raised without antibiotics” or “no antibiotics ever”. Finally, it cannot be ruled out that resistance to ionophores might induce cross-resistance to human health care relevant antibiotics, although there is no conclusive proof and it is generally deemed very unlikely.
4. How prevalent is antibiotic use in poultry production?
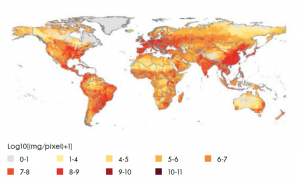
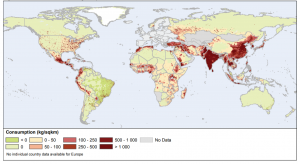
Globally, limited quantitative data exists on antibiotic use in livestock. Van Boeckel et al. (2015) have built statistical models based on livestock densities, projections of demand for meat products and current estimates of antimicrobial consumption in high-income countries to model global use of antibiotics for 2010 and 2030. For 2010, they estimate the global average annual consumption of antimicrobials per kilogram of animal produced was 45 mg/kg, 148 mg/kg, and 172 mg/kg for cattle, chicken, and pigs, respectively. Figure 1 depicts antimicrobial consumption in livestock worldwide.
Figure 1. Global antimicrobial consumption in livestock in milligrams per 10 km2. Source: Van Boeckel et al., 2015
Global trends in antimicrobial use in food animals. Van Boeckel et al. PNAS May 5, 2015. 112 (18) 5649-5654; https://doi.org/10.1073/pnas.1503141112
5. Is antibiotic use in livestock production expected to grow?
Globally, consumption of antimicrobials in food animal production is projected to increase 67% by 2030 compared to 2010 levels. In Asia, antimicrobial consumption is projected to reach 51,851 tons by 2030, representing 82% of global antimicrobial consumption in 2010. Starker increases in antibiotic consumption in some regions compared to others are largely attributable to a shift toward more intensive production systems as well as increased demand for animal protein caused by population and income growth in these regions. Figure 2 depicts projected increased growth in demand for poultry meat from 2000 to 2030.
Figure 2. Growth in demand for poultry meat from 2000 to 2030 (in ‘000 MT). Source: FAO 2011
Source: Food and Agriculture Organization of the United Nations, 2011, T.P. Robinson & F. Pozzi, Mapping supply and demand for animal-source foods to 2030], http://data.fao.org/ref/518d1321-255b-4b0b-ab53-f55cba1d8ad2.html?version=1.0. Reproduced with permission
Many governments are introducing legislation to support antibiotic-reduction initiatives. For decades, the European Union has committed to reducing antibiotics. In some countries such as the Netherlands, antibiotic susceptibility testing is mandatory when prescribing third choice, critically important antibiotics. Currently, the European Union is taking steps to prohibit the prophylactic group treatment of animals. The U.S. Veterinary Feed Directive, enacted in 2017, requires a veterinarian’s authorization to purchase antimicrobials deemed medically important. China banned colistin as a feed additive in 2016 and is now introducing legislative actions to reduce antibiotic use in general and ban in-feed antibiotics by 2020. Indonesia and Vietnam banned the use of antibiotics for growth promotion starting in January 2018. These are a few selected examples to illustrate the truly global attention to the topic. Next to governmental actions, the private sector is also moving in the same direction, often going far beyond legislation. Major U.S. brands, for example, have initiated various production schemes ranging from responsible antibiotic use to completely antibiotic-free production.
6. Will reducing antibiotics use negatively impact animal health and production profitability?
Reducing antibiotic use should not compromise animal health and welfare. Focus should be on reducing the need for antibiotics, the reasoning being that a healthy animal will not need antibiotic treatment. In this context, it is important to realize that overuse of antibiotics and subsequent resistance will not only affect human health care but will also limit treatment options for our animals. It is in the poultry producers’ direct interest to maintain the efficacy of antibiotics by limiting their use.
There is no quick-fix solution, no simple “replacement for antibiotics”. Management mistakes that are to some extent covered-up by antibiotics need to be tackled. In practice, we therefore sometimes even see an improved health and performance when reducing antibiotics. It is more complex, of course, to eliminate antibiotics completely, especially when ionophores are included in the definition of “antibiotic-free”. Our experiences with integrators leading in antibiotic reduction have demonstrated it is possible to produce broilers without antibiotics (including ionophores) with comparable or even improved health and performance (with about only 1-2% of all flocks needing antibiotic treatment) and greatly improved profitability.
7. I want to reduce or even eliminate antibiotics from my production. What should I do?
There is good reason to reduce antibiotic use in poultry production. Beyond addressing the antibiotic resistance risk and maintaining the efficacy of antibiotics for both humans and animals, reducing antibiotic use can add value to the end product and increase your profitability. Reducing or even eliminating antibiotics from your poultry production will require an integrated approach. You must be willing to embark on a journey that includes reviewing your feed formulation, water management, farm practices and health management. Feed and water additives provide important support in antibiotic-reduction programmes but need to be accompanied by best practices across the production chain. Starting in the breeder flock, continuing to the hatchery and finally the broiler farm, including the feed mill with its production processes and raw material management. Trouw Nutrition’s feed-farm-health-programme encompasses detailed analyses of the critical control points in the entire poultry production chain. Based on these analyses, tailor-made solutions are designed for each producer’s situation. There is no panacea; points of special attention will differ for each producer, as will the solutions. This is how Trouw Nutrition aspires to honor our commitment: to enable customers to reduce the need for antibiotics while maintaining profitability.
References available upon request.